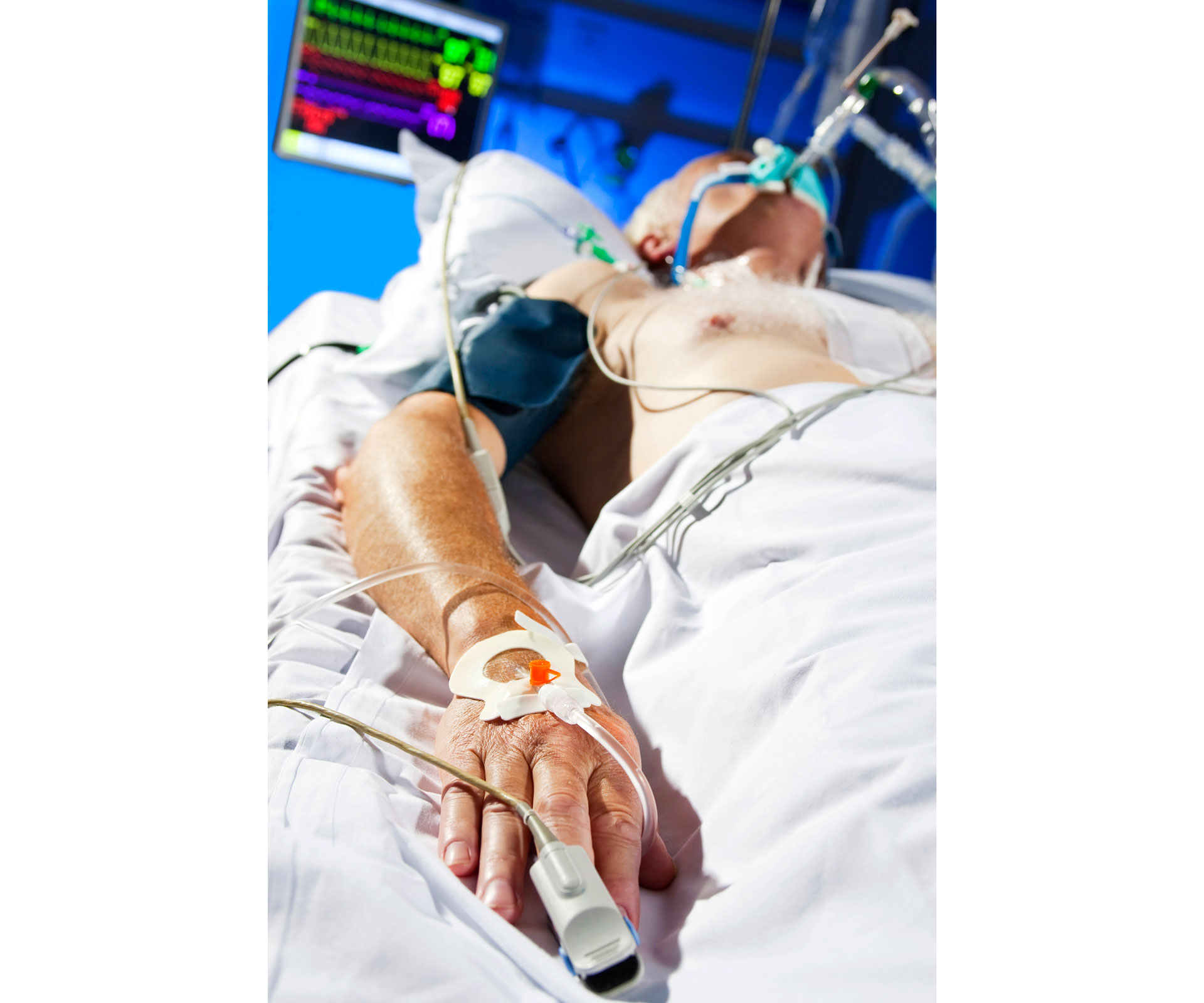
When you’re wheeled into the intensive care unit with your life-threatening illness, pretty often you’ll get what’s known as a central venous line catheter put in you. This is not bladder-related, so relax. But don’t relax too much.
What happens is a doctor will stand over your shoulder and very gently but firmly and terrifyingly push a quite large-gauge needle indeed into your upper chest, and right down into a vein leading directly into one of the great vessels of your heart, leaving a wire in there.
She’ll then gently run a catheter over that wire, sliding it down inside those big thick veins, so the tip of the catheter sits snugly right next to your dark, undeserving heart where parenteral nutrition and fluids and drugs can be fed in rapidly and efficiently as your condition, fluctuating wildly, demands. You’re in an ICU, after all, and things are already pretty dicey.
The end of this catheter, hanging outside like some weird brooch, is a little octopus of tubes and taps, sewn and taped to your chest. These get infected all the time.
And the infection goes straight into your bloodstream, unprotected as it is by the best infection control device there is: skin. Ten to 50 per cent of people who get their bloodstream infected by a central line may flat out die because of it. Fevers, shaking, delirium, multi-organ systemic failure. Costs to the hospital of $20,000 to $50,000 each. And ICUs, since forever, have just accepted that this is what comes with the territory with a central line catheter.

Until along came a tan, boyish, blond doctor at Johns Hopkins Hospital in Baltimore named Peter Pronovost.
In 2006, Pronovost published a study in the New England Journal of Medicine (basically the New York Times of medical journals) showing how in his ICU at Johns Hopkins he’d demonstrated that what you need to do is five little things on a checklist and boom: you can reduce catheter-related bloodstream infections in your ICU to zero.
None. But you have to do those five things, and you have to do all five and you can’t leave any out. They call it a “bundle”. He did it first at Johns Hopkins, then in 120 ICUs in Michigan. In a year and half, they saved 1500 lives.
It blew everybody’s minds, but it blew them slowly – no one wanted to do it anywhere else for years. But in time people began to realise: doctor as hero, as priest, doesn’t work. Teamwork, checklists: they work. What happened became known in medical shorthand as “what happened in Michigan”, or just “Michigan”. The UK’s National Health Service went all out to imitate it – its programme was actually called “Matching Michigan”. In 2008, Pronovost was made a MacArthur Fellow and one of Time’s top 100 people of the year.

US intensive care specialist Peter Pronovost. His work has “already saved more lives than that of any laboratory scientist in the past decade”, according to Atul Gawande in The New Yorker.
That same year, Mary Seddon, a doctor and “quality improvement” manager at Auckland’s Middlemore Hospital with an obsession for quality and safety – and a certain tense rigour and disdain for needless smiling or approachability that has earned her a rather frightening nickname – took Pronovost’s idea to Pat Mead, and Catherines Hocking and Simpson, then Middlemore’s head of infection prevention and control, ICU quality facilitator, and ICU clinical head, respectively. Within two years, they’d published their study in our version of the New England journal, the New Zealand Medical Journal: bloodstream infections at Middlemore reduced to zero.
What nobody outside the healthcare sector knows is New Zealand is the first country in the world to take one of the most ground-breaking – but ridiculously simple – advances in hospital-acquired infection control in the 21st century and within two years as a nation almost eradicate the brutal, killer infections of the bloodstream caused by central line catheters in the desperately sick people in our intensive care units.
It started at Middlemore and went national. We’re saving hundreds of lives and millions of dollars and we did it like no other country has been able. Without any governmental regulation or decree, arising entirely out of good will, good faith and good science.
The person who started it all is known throughout New Zealand medicine as “Scary Mary”.

Mary Seddon “Scary Mary”
I interviewed Dr “Scary” Mary Seddon, 51, mother of one, now in Queensland, Australia, by video. Scary Mary has a reputation. Her iPad’s cover-stand apparatus wasn’t working, so she’d laid the tablet on her desk. On my screen, the impression, before anything of substance was even said, was already of an impatient, narrow, short-haired, bespectacled intensity, looming over me.
“Can you tell me,” I started out asking, as warmly as I felt able, “about your early years, school, college?”
Pause. “Really?” She audibly sighed.
The thing is, doctors don’t always do the same things – even when confronted with the same situation, the same symptoms, the same diagnoses in broadly comparable patients. Doctors can’t necessarily keep up with the flood of medical knowledge constantly produced and published worldwide. They develop their own ways, their own favoured solutions to problems. A surgeon might administer a particular antibiotic before a procedure because he’d heard about it at a conference the previous year. A doctor might insert a central line as he’s always done, in rubber gloves and a cap but without covering the patient with a sterile drape, because he was taught that way. Practice diverges.
But in certain medical situations, standardisation and “factory floor” medicine are clearly desirable – routine ops, for example, frequently performed, so there is a huge amount of data to prove what works best. There is a Canadian hospital that specialises in hernia repair operations, by sewing together overlapping muscle layers under just a local anaesthetic. They fix more than 7000 hernias a year, almost 20 a day. And that’s all they do. Their 99.5 per cent lifetime success rates are the highest in the world, which – the rest of the world – typically puts up with 10 to 25 per cent of repaired hernias popping open again.
Who was Scary Mary? The interview was not going well. The pause lingered long. “Um, really,” I said.
“I’m from a big family, number six of seven,” she said, looming over me. “Five girls, two boys. It took a long time to get a room of your own. Born in Tauranga, went to all the local schools. Dad was a GP, Mum was a homemaker who became a city councillor after the kids left home.”
I was listening closely. She had a clipped, reluctant delivery; guarded, but what was behind it? Why was she scary? Was she hard, bitter? Or just hard-bitten? Medicine’s a notorious boys’ club. I asked her about gender at med school. She said it wasn’t an issue.
“When you’re young you think anything’s possible.” She laughed slightly, for the first time.
“Do you still feel that way?” I asked.
“Nah…”
I flicked through Mary Seddon’s CV: University of Otago, Bachelor of Medicine, Bachelor of Surgery, with distinction. “I liked science and arts at school,” she said, “and medicine seemed a good mix of the two. It seemed probably the most difficult thing to do, so that attracted me, I guess.”
Every undergraduate prize and distinction and scholarship you can think of. Medical registrar roaming over the specialties, all in the Auckland area, then Tauranga.
Master of Public Health, University of Auckland, first-class honours. Cardiovascular research fellow. Then the incredibly prestigious and hard-to-get Harkness Fellowship, which she shared with Ron Paterson, future NZ Health and Disability Commissioner, and which took her to Harvard Medical School. Consultant back at Middlemore. Fellow of the Royal Australasian College of Physicians.
A gleaming career, clearly. She was on a specialist-physician fast track, but then suddenly this move back to Tauranga, and then to public health, in 1995. I figured it out.

Dr Mary Seddon with her daughter, Kate, 20, a medical student at Otago.
In 1994, Seddon had a daughter, Kate, now 20 and at med school at Otago. Seddon was a solo mum in her last year of registrar training, and if you don’t know what’s involved with that, it means at least 12-hour days. It means 7am meetings. It means nights. It means on-call whenever. And that’s the job, not the parenting of a one-year-old. She moved back to Tauranga, and, with her parents’ help with child-minding, finished the last year of her registrar training and began her master’s degree in public health. She basically never stopped.
“Doing my master’s, I got into the routine of having Kate in bed by 8pm, getting to my desk by 8.30 and working till midnight. I finished it three days before we left for the US. Kate was three and a half. Harvard daycare had a two-year waiting list, so I found a place over the train tracks from Harvard School of Public Health. It was a poor area and she was the only white kid. But kids are colour-blind. It just meant I had to put a lot of plaits and coloured beads in her hair because all the other kids had such elaborate cornrows.”
But back to 2008. A quality improvement manager at Middlemore. What is “quality improvement”? What’s this weird, opaque noun “quality”? In the past 20 years, worldwide and here, there’s been an obsession with health care and hospitals making health care just straight up better. Doctors, nurses and managers have been working, studying and publishing on ways to make sure fewer people are harmed in our hospitals, through sponges and instruments left in after operations, through the wrong drugs or too much of a drug being administered.
Working on ways of making health care more efficient, and more open to more people. On saving money and directing it to places where it’s needed. On making practice more uniform where it ought to be uniform. It’s a science and an art all its own. And they’ve been working on reducing the infections that people get in hospitals.
At Middlemore, they set about figuring out exactly how many central line infections were actually occurring, and establishing a baseline to see if what they were going to do would have any effect. Seddon, Mead, Hocking and Simpson went through 18 versions of the checklist they’d designed to ensure their nurses and doctors did everything required by the bundle. Sometimes, they found, steps were skipped when equipment wasn’t available, the trolley wasn’t restocked, or crucial stuff was scattered around the ICU.
So they designed a sterile pack containing everything: the full gown and hat, the gloves, drapes to cover the patient completely, the syringes and the needles. It opened up, revealing – like pass the parcel or origami – each piece of equipment in sequence, as needed. They were trying to make it easier to follow the bundle than not.“The proceduralist would have to actually pick something up and throw it away not to follow the bundle and they wouldn’t do that,” said Hocking.
“We had clinicians who didn’t want to comply with it,” said Seddon. “There was one department that just saw this as cookbook medicine and didn’t want anything to do with it. But we just got started.”
The pack cost $80 a pop. They were trying to make the right thing the easiest thing to do.
Seddon, Hocking and Simpson took the work to doctors and infection control nurses at Auckland Hospital. Then, in October 2011, Ko Awatea (“dawning of the first light”), the quality improvement and innovation centre at Counties Manukau Health, entered into a partnership with New Zealand’s national health quality improvement body, the Health Quality & Safety Commission.
There are 20 district health boards in this country, with 25 ICUs and about 19,000 ICU patients admitted every year. Hawke’s Bay’s ICU alone, for example, places approximately 400 central lines in patients every year. Was it feasible? Many of these ICUs weren’t even collecting data on the number of days patients were on central lines. They had different definitions for what was an infection and what wasn’t; different ways of doing their blood cultures; even different luer locks (joining needles to syringes) on their catheter taps. There were dozens of problems, and dozens of top-of-the-food-chain, Type A personalities working in our hospitals to convince of the merits of the idea.
But given the evidence the bundle itself worked, said Seddon, “it didn’t seem ethical to delay”. The Health Quality & Safety Commission brought in key generals, heavy-hitters like infection control expert Sally Roberts and ICU clinical leader Shawn Sturland, to run the tactics on the ground. They recruited; they converted. They called it a “collaborative”. They divided up the country in order to conquer. They estimated baselines. They shared the pack, the how-to guide, and everything they knew. And they went national.
“Never doubt that a small group of thoughtful, committed citizens can change the world. In fact, it is the only thing that ever has.” – anthropologist Margaret Mead

Within two years, bloodstream infections caused by infected central line catheter insertions at Middlemore Hospital had been reduced to zero.
Seddon quotes quality improvement expert Paul Batalden in her 2011 study, who says, “‘Every system is perfectly designed to produce the results that it gets.’ Prior to this initiative the central line insertion system in the ICU at Middlemore Hospital was perfectly designed to harm 10 to 14 patients a year through central line infections.
“In quality improvement,” she told me, “it’s all about establishing if there is a problem. Some didn’t think their rate of just over six infections per 1000 line days was a problem. We had a central line infection once or twice every month and that was just the way it was. That’s why the Michigan paper was so important.” The slightest of pauses. “Because we could show that it didn’t need to be that way.”
Her voice was changing. In the way she’d said that word – “need” – I heard a softening, a kindness, even something that sounded like, well, pain.
“Health care is a sacred mission,” the Lebanese-American father of health quality assurance Avedis Donabedian said a month before he died. “Doctors and nurses are stewards of something precious.” Here, a $49 pack of equipment (by 2013: bulk-buy power) and a certain intense focus on the way we do things and that’s someone’s life saved, and $20,000 to $50,000 to spend on something else important, like oncology, like the neonatal ward. In total, the Health Quality & Safety Commission has estimated the programme has saved the country more than $4 million in a little over two years.
“Is it important to you to be excited?” I asked Seddon.
“Absolutely. Quality improvement is hard. You need to have tasks where you think you’re going to make a difference.” Then she said something surprising to me. “Not being a clinician anymore, not looking after patients, this is the next best thing I can do.”
There was real sadness there. “The ‘next best thing’?”
“Kate has been the finest thing that has happened to me, but when she was growing up it was just too difficult. I worked about five months each winter to help with the winter load, the rest of the year in quality improvement. But trying to get after-school care, before-school care, was too hard. I couldn’t come in at night. So in the end I elected to go fulltime in quality improvement. I may go back to looking after patients one day.”
The steel in her voice had returned. “It’s hard to know.”
Speaking of steel, I now prepared myself to ask the question I’d been dreading and prepping myself for the entire interview. About that nickname.
Short, sharp exhalation. Followed by a long pause. I didn’t know where to look. Did she in fact even know about it? I didn’t know if she knew. What if I were the first to tell her? Maybe she’d admire my daring and not slam the iPad down on me? Maybe she’d feel it was in the public interest?
“I got that,” she said at last, and her voice was calm, “when I was a medical registrar. I do expect high standards. I do tend to speak before I think. My bark is certainly worse than my bite and it’s not a nickname of which I’m necessarily proud.” There was a small smile. “I don’t think I’m scary anymore. Now I call myself ‘Postpartum Mary’.”
I apologised. “I had to ask.” She verifiably laughed.
It took a long time to get a room of your own, she’d said of her big family, and perhaps one day she will return to New Zealand. Mary Seddon both is and is not a patient person. But meanwhile the legacy of the programme is in place. ICUs all over the country are connected – they know who’s doing what and what works. They’re collecting the right data, and crucially the same kinds of data, so they can prove that it’s working. Recent figures published by the Health Quality & Safety Commission show nationwide central line infections remain at less than one per thousand days of central lines inserted. That’s zero.
Ten to 50 per cent – those who die from their central line infections – of zero is zero.
The arithmetic is both simple and profound.